- Clone
- A15088B (See other available formats)
- Regulatory Status
- RUO
- Other Names
- Ciliary Neurotrophic Factor, HCNTF
- Isotype
- Mouse IgG2b, κ
- Ave. Rating
- Submit a Review
- Product Citations
- publications
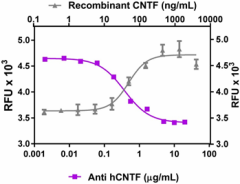
-

Recombinant human CNTF (grey triangles, Cat. No. 559802) induces the proliferation of TF-1 human erythroleukemic cells. Anti-human CNTF antibody (clone A15088B, purple squares) inhibits the effect of recombinant human CNTF (at 400 ng/mL) in TF-1 cells in a dose dependent manner. The ND50 = 0.2 – 1.0 µg/mL.
| Cat # | Size | Price | Quantity Check Availability | Save | ||
|---|---|---|---|---|---|---|
| 608503 | 100 µg | $270 | ||||
| 608504 | 1 mg | $715 | ||||
CNTF, ciliary neurotrophic factor, was initially identified as a survival factor for cultured embryonic chick parasympathetic neurons. The human protein has 84% similarity to the rat and rabbit proteins. It lacks an N-terminal signal sequence and consensus sequences for glycosylation or hydrophobic regions. The CNTF profile shows similar sequence and structure motifs to hematopoietic cytokines (IL-6, IL-11, LIF, oncostatin M, cardiotrophin-1) and thus was considered a member of the IL-6 family. CNTF exerts effects on glial cells and neurons. CNTF activates astrocytes, which promote their capacity to support neurons and oligodendroglia. CNTF stimulates astrocytes to secrete FGF-2 and rat microglia to secrete glial cell line-derived neurotrophic factor (GDNF). CNTF is produced by astrocytes after brain injury. The CNTF receptor complex includes the CNTF binding protein (CNTFRα), the LIF binding protein (LIFRb), and the signal transducer of IL-6 (gp130). CNTFRα does not have transmembrane or intracellular domains, which means it does not induce signal transduction per se. CNTFRα is linked to the cell membrane via glycosyl phosphatidylinositol linkage, and it can be released by phospholipase C to produce a soluble receptor. The complex formed by CNTFRα and CNTF serve as agonists for cells that do not express CNTFRα. Cardiotrophin-like cytokine (CLC) also binds to CNTFRα, which is proposed to activate gp130, which are LIFRb subunits. CNTF reduces the symptoms of experimental autoimmune encephalomyelitis, and its absence exacerbates the severity of multiple sclerosis disability. CNTF has been studied in retinal degenerative disorders and was shown as a potential clinical application.
Product DetailsProduct Details
- Verified Reactivity
- Human
- Antibody Type
- Monoclonal
- Host Species
- Mouse
- Immunogen
- Recombinant human CNTF
- Formulation
- Phosphate-buffered solution, pH 7.2.
- Preparation
- The anti-human CNTF antibody was purified by affinity chromatography.
- Concentration
- The antibody is bottled at the concentration indicated on the vial, typically between 2 mg/mL and 3 mg/mL. Older lots may have also been bottled at 1 mg/mL. To obtain lot-specific concentration and expiration, please enter the lot number in our Certificate of Analysis online tool.
- Storage & Handling
- The antibody solution should be stored undiluted between 2°C and 8°C. This Ultra-LEAF solution contains no preservative; handle under aseptic conditions. Do not freeze.
- Application
-
Neut - Quality tested
- Recommended Usage
-
Each lot of this antibody is quality control tested by blocking the inhibition of proliferation induced by human recombinant CNTF (400 ng/mL) on TF‑1 human erythroleukemic cells. ND50 range: 0.2 – 1.0 mg/mL. It is recommended that the reagent be titrated for optimal performance for each application.
- RRID
-
AB_2783353 (BioLegend Cat. No. 608503)
AB_2783354 (BioLegend Cat. No. 608504)
Antigen Details
- Structure
- Homodimer
- Distribution
-
Astrocytes, microglia, hepatocytes, skeletal muscle, embryonic stem cells, and bone marrow stromal cells.
- Function
- Stimulates gene expression, along with cell survival or differentiation in neuronal cell types. It is a potent trophic factor for motor neurons. IL-1b, TNFα (TNF-α), and EGF induces the release of CNTF from astrocytes.
- Interaction
- Sensory, sympathethic, ciliary, motor neurons, microglia, retinal ganglion cells, adipocytes, and skeletal muscle cells.
- Ligand/Receptor
- Trimeric complex form by CNTF receptor-α(CNTFRα), LIF receptor (LIFR), and gp130/ CD130.
- Cell Type
- Astrocytes, Embryonic Stem Cells, Microglia
- Biology Area
- Cell Biology, Stem Cells
- Antigen References
-
- Negro A, et al. 1991. Eur. J. Biochem. 201:289.
- Bazan JF. 1991. Neuron 7:197.
- Davis S, et al. 1993. Science 260:1805.
- Plun-Favreau H, et al. 2001. EMBO J. 20:1692.
- Lin HW, et al. 2009. J Neuroinflammation 6:7.
- Wen R, et al. 2012. Prog. Retin. Eye Res. 31:136.
- Gene ID
- 1270 View all products for this Gene ID
- UniProt
- View information about CNTF on UniProt.org
Related Pages & Pathways
Pages
Related FAQs
- Do you guarantee that your antibodies are totally pathogen free?
-
BioLegend does not test for pathogens in-house aside from the GoInVivo™ product line. However, upon request, this can be tested on a custom basis with an outside, independent laboratory.
- Does BioLegend test each Ultra-LEAF™ antibody by functional assay?
-
No, BioLegend does not test Ultra-LEAF™ antibodies by functional assays unless otherwise indicated. Due to the possible complexities and variations of uses of biofunctional antibodies in different assays and because of the large product portfolio, BioLegend does not currently perform functional assays as a routine QC for the antibodies. However, we do provide references in which the antibodies were used for functional assays and we do perform QC to verify the specificity and quality of the antibody based on our strict specification criteria.
- Does BioLegend test each Ultra-LEAF™ antibody for potential pathogens?
-
No, BioLegend does not test for pathogens in-house unless otherwise indicated. However, we can recommend an outside vendor to perform this testing as needed.
- Have you tested this Ultra-LEAF™ antibody for in vivo or in vitro applications?
-
We don't test our antibodies for in vivo or in vitro applications unless otherwise indicated. Depending on the product, the TDS may describe literature supporting usage of a particular product for bioassay. It may be best to further consult the literature to find clone specific information.
Other Formats
View All CNTF Reagents Request Custom Conjugation| Description | Clone | Applications |
|---|---|---|
| Ultra-LEAF™ Purified anti-human CNTF | A15088B | Neut |
Compare Data Across All Formats
This data display is provided for general comparisons between formats.
Your actual data may vary due to variations in samples, target cells, instruments and their settings, staining conditions, and other factors.
If you need assistance with selecting the best format contact our expert technical support team.
-
Ultra-LEAF™ Purified anti-human CNTF
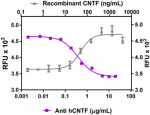
Recombinant human CNTF (grey triangles, Cat. No. 559802) ind...
 Login/Register
Login/Register 








Follow Us